|
This last equation provides a means of measuring work functions of different materials. Conversely, if the light frequency is decreased below the threshold value f 0, the photons will not have enough energy to overcome the work function at the threshold frequency f 0 photons would be released but with zero kinetic energy, so that However, increasing the light frequency will increase the photon energy, resulting in more energetic photoelectrons (Fig. Increasing the light intensity increases the number of photons per second arriving at the metal electrode but does not change the energy of each photon, so the photoelectron energy K max and the stopping voltage V s remain the same (Fig. Values range from 2.2 eV (for the alkali metal lithium) to 6.35 eV (for the noble metal platinum).Įinstein's concept explains the two puzzling features of the photoelectric effect. 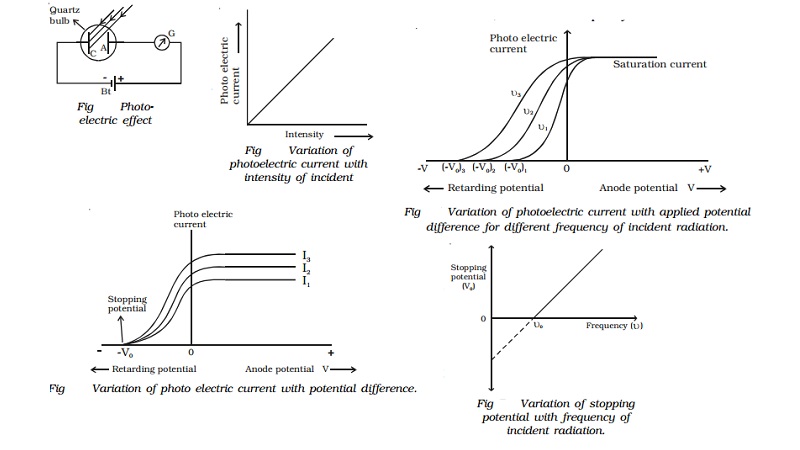
For convenience, this work function is normally measured in units of electronvolts (eV): 1 eV = 1.6 ´ġ0^-19 J. Where phi is the work function of the metal it represents the minimum energy which must be supplied to release an electron from the metal. If this process occurs at the metal surface, the electron is released into the vacuum with a kinetic energy given by: In the photoelectric effect, each photon donates all of its energy hf to an electron in the metal. Įxtending this idea to all cases of electromagnetic waves, the energy of the radiation is present in "bundles" see Fig.2.16b. 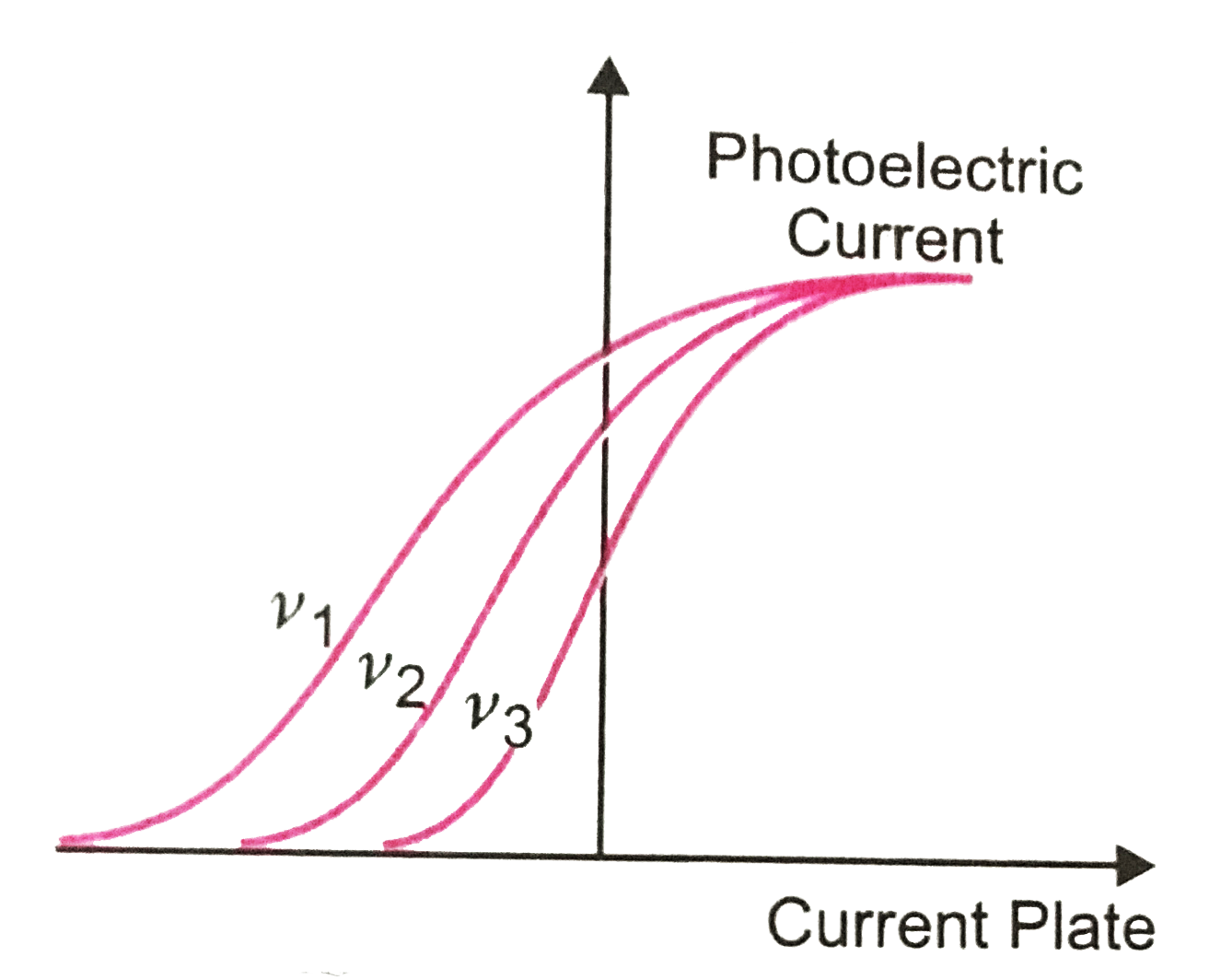
Using ideas derived from thermodynamics, he showed that cavity radiation could be treated as a system of particles, which we now refer to as photons, each carrying a quantum of energy of magnitude hf. Einstein had recognized an inconsistency between Planck's idea of quantization of oscillators (in the walls of a cavity) and the usual assumption that cavity radiation consists of continuous electromagnetic waves. In fact, his explanation of the photoelectric effect ( not the more controversial theory of relativity) was cited as his greatest contribution to physics when he was awarded the Nobel Prize in 1922. In one of his papers published in 1905, Einstein came up with an explanation for these discrepancies. For a given light intensity, we would expect the electric field E to be independent of frequency or wavelength, on the basis of classical physics. It is this electric field which is presumed responsible for attracting electrons out of the metal into the vacuum and larger E should result in electrons with higher kinetic energies, requiring a larger negative V s to stop them reaching the collector.Īlso at odds with classical theory is the fact that V s (or K max) varies with frequency f of the incident light in fact the experimental data lie on a straight line with intercept f 0 see Fig. According to classical theory, larger I implies a stronger electric (and magnetic) field E in the incident electromagnetic wave. What cannot be explained on the basis of classical physics is the fact that the value of V s is independent of the light intensity I. At that point, even the most energetic photoelectrons (those released with a kinetic energy K max, assuming there is some spread in kinetic energy K) will exchange all of their kinetic energy for potential energy eV s by the time they reach the collector, in other words K max = V s. This behaviour can be explained in terms of the negative collector repelling the photoelectrons, reducing the photocurrent or suppressing the photocurrent entirely (by stopping the electrons reaching the collector) when V reaches the stopping voltage V s. When the battery leads were reversed so that the applied bias V was negative, the photocurrent decreased and became zero if the magnitude of the negative bias exceeded a certain value V s. When the light intensity was increased, the photocurrent also increased, indicating that more photoelectrons were being produced. This is easily explainable in terms of the positive collector electrode attracting a larger fraction of emitted negatively-charged electrons. 2.15a.įor a given intensity I of the ultraviolet light, the photocurrent increased with increasing positive voltage. By adjusting the potentiometer setting so as to vary the voltage V applied to the "collector" electrode, the photocurrent A could be plotted against applied voltage, as in Fig. In 1902, the German physicist Philipp Lenard started to investigate this photoemission phenomenon in detail, using an apparatus involving two electrodes in a vacuum tube, as shown schematically in Fig.  
This was convincing evidence that atoms contain electrons. In the 1890's, it was discovered that metal surfaces in vacuum which are exposed to ultraviolet light emit electrons. Figure references are to the first edition of Modern Physics by Serway, Moses and Moyer (Saunders, 1989).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
